Role of O-linked Glycans in Animal Development and Notch Signaling
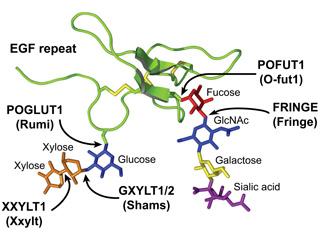
The Notch signaling pathway plays important roles in animal embryonic development and adult maintenance. We and others have shown that addition of O-linked glycans (carbohydrates) to Notch EGF repeats regulates various aspects of Notch signaling in flies and mammals. Notably, we have found that in some contexts, the Notch pathway is sensitive to the gene dosage of the enzymes responsible for the addition of the xylose-xylose-glucose-O glycans to EGF repeats.
Moreover, our recent report on the identification of a hypomorphic POGLUT1 allele in patients with a new form of limb-girdle muscular dystrophy (LGMD-2Z) indicates that myogenesis is highly sensitive to Notch glycosylation by POGLUT1.
Our current studies are aimed at elucidating the molecular bases for tissue -specific regulation of Notch signaling by xylose-xylose-glucose-O glycans. Another major goal is to understand how the corresponding glycosyltransferases regulate Notch signaling in a dosage-dependent manner.
These studies might help us establish a framework for therapeutic modulation of Notch pathway in diseases caused or exacerbated by aberrant Notch signaling.
Genetic Mechanisms for Biliary Development and Repair, with a Focus on Alagille Syndrome
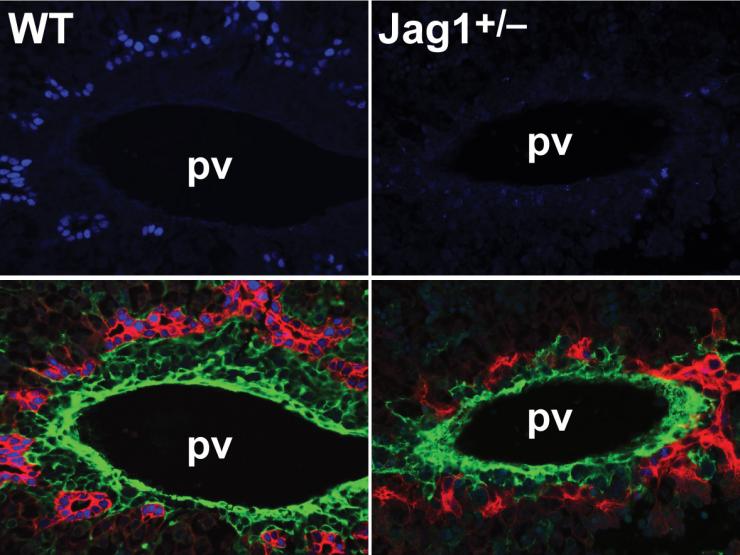
Alagille syndrome (ALGS) is an autosomal dominant disorder characterized by a congenital cholangiopathy of variable severity accompanied by cardiac, skeletal, renal and other abnormalities. In 94% of cases, ALGS is caused by mutations in JAG1, which encodes one of the ligands for the Notch pathway.
We have recently reported a mouse model for ALGS and have identified Poglut1 as a dominant genetic suppressor of the ALGS biliary phenotypes. Ongoing experiments are aimed at using this model to better understand the pathophysiology of ALGS and to determine the basis for its extreme phenotypic variability, even among patients harboring the same JAG1 mutation. We have also identified a transcription factor and another glycosyltransferase which modify the Jag1[+/–] phenotypes in a dosage-sensitive manner. We are taking advantage of this mouse model and its genetic modifiers as a framework for identifying mechanism-based therapies for ALGS.
Role of Deglycosylation in Animal Development and Human Disease
Patients with mutations in N-glycanase 1 (NGLY1) exhibit a host of developmental abnormalities including a delay in physical and intellectual development, movement disorders, chronic constipation, osteopenia and lack of tears.
NGLY1 is a cytoplasmic deglycosylation enzyme capable of removing N-linked glycans from N-glycoproteins. It has been proposed that upon retrotranslocation of misfolded N-glycoproteins from endoplasmic reticulum (ER) to cytosol, NGLY1 cleaves the N-glycans to facilitate the proteasomal degradation of misfolded proteins. However, the biologically-relevant targets of NGLY1 that contribute to the patient phenotypes and the developmental processes sensitive to loss of its activity in animals are not well understood.
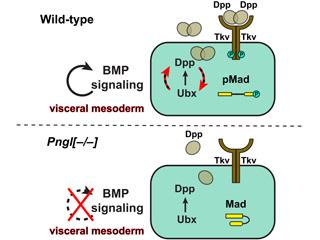
Using Drosophila, we have discovered that NGLY1 (known as Pngl in flies) specifically regulates the bone morphogenetic protein (BMP) signaling during Drosophila intestinal development. In addition, our work suggests that Pngl regulates several other aspects of Drosophila development independently of its role in BMP signaling.
Using a multidisciplinary approach, we are studying the cellular and physiological defects caused by the loss of Pngl in flies. We are also examining whether our findings in flies can shed light on the function of the mammalian NGLY1, with the goal of identifying potential therapeutic targets for the disease.








