Prosthetic Replacement of the Ocular Surface Ecosystem (PROSE) is a pioneering treatment model developed by Boston Foundation for Sight (BFS) to restore vision, support healing, reduce symptoms, and improve the quality of life for patients suffering with complex corneal disease. PROSE uses an FDA-approved custom-designed and fabricated prosthetic device to replace or support impaired ocular surface system functions that protect and enable vision.
What is the PROSE Device?
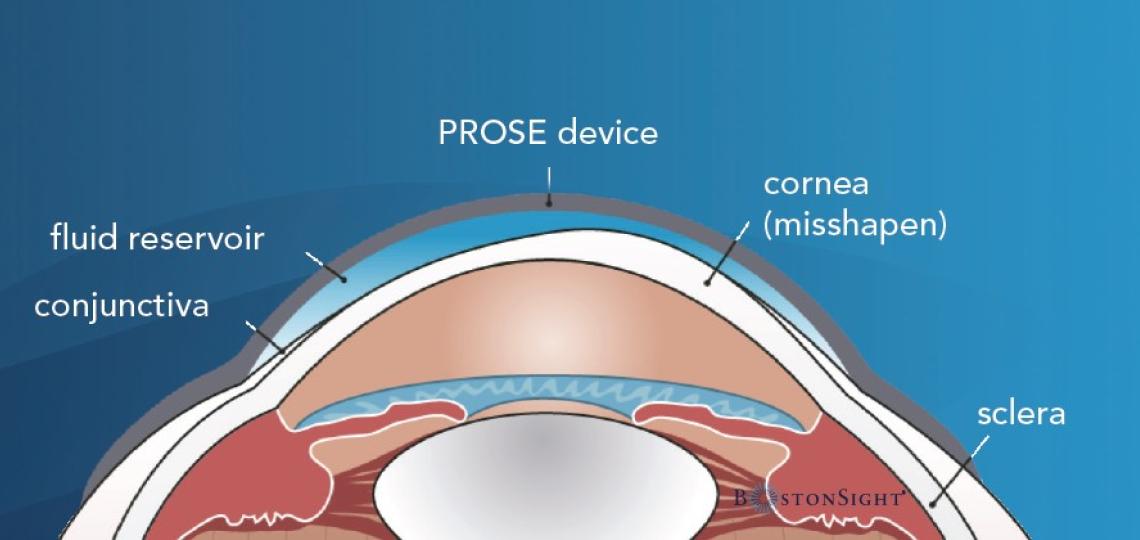
The PROSE device is a transparent dome made of an oxygen permeable plastic that rests on the white part of the eye, sclera, and vaults over the cornea. It is filled with a preservative-free saline and inserted in the eye, which provides constant lubrication to the cornea. It provides a smooth surface over an irregular, damaged, or diseased cornea which can promote visual function and improve ocular comfort.
The PROSE Device for Ocular Surface Diseases
Ocular surface disease (OSD) can be caused by a multitude of etiologies and, when severe, can lead to debilitating symptoms. Some conditions that may benefit from PROSE treatment are, but not limited to, keratoconus, corneal transplantation, LASIK complications, Stevens-Johnsons Syndrome, and Sjogren’s syndrome.
Steps in the Treatment Process
- Referral from primary eye care provider, medical doctor, or cornea specialist.
- Initial medical review
- Determination of insurance coverage and potential eligibility (3-6 weeks on average)
- Consultation with a PROSE Clinical Fellow to assess candidacy.
- PROSE treatment (5-10 day-long visits).
- Periodic follow-up visits as required during the first 6 months.
- Annual evaluation of PROSE fit annually.
Patients are recommended to continue follow-up care with their primary eye care provider throughout treatment, and ongoing care is coordinated with primary providers and the PROSE treatment team.
If you are a referring provider, please fill out this PROSE referral form and fax it to 713-798-8769.
Call 713-798-6109 today to learn more or set up a consultation.








